
In order to know the position of an object, we throw the photons of light upon them. Heisenberg has given a principle in this connection. But this is not possible for electron, proton, and neutron which are microscopic particles. If we know its initial position and momentum, then we can predict its position and momentum at any other time. When we are studying a large moving object say a planet, then we can follow its definition path on which it travels.
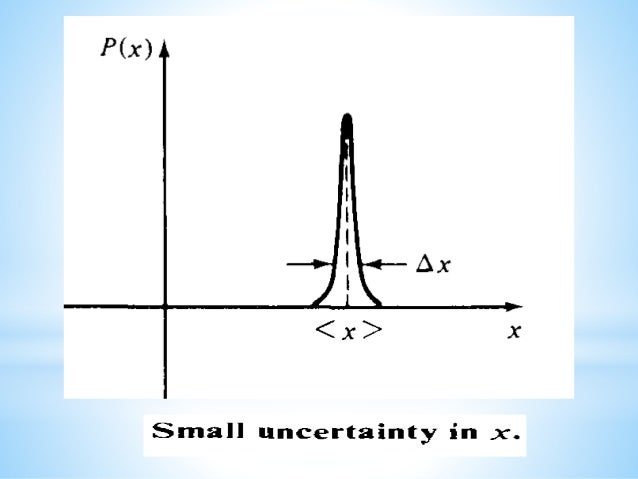


The uncertainty principle is defined as: “It is impossible to measure simultaneously both the position and momentum of a microscopic particle with accuracy or certainty.” Keep reading Heisenberg uncertainty principle Statement

 RSS Feed
RSS Feed
